DigitalCabinet is built and owned by Ethlen. Aretetic Solutions holds the exclusive US license.
Launch in Weeks, Not Years
Most organizations spend years navigating IRB approvals, infrastructure procurement, and custom software development before enrolling their first participant. DigitalCabinet eliminates that entirely.
We provide a baseline IRB-approved consent framework that covers core data collection and storage requirements — meaning your program can launch without starting from scratch on ethics review. Your team focuses on the science. We handle the platform.
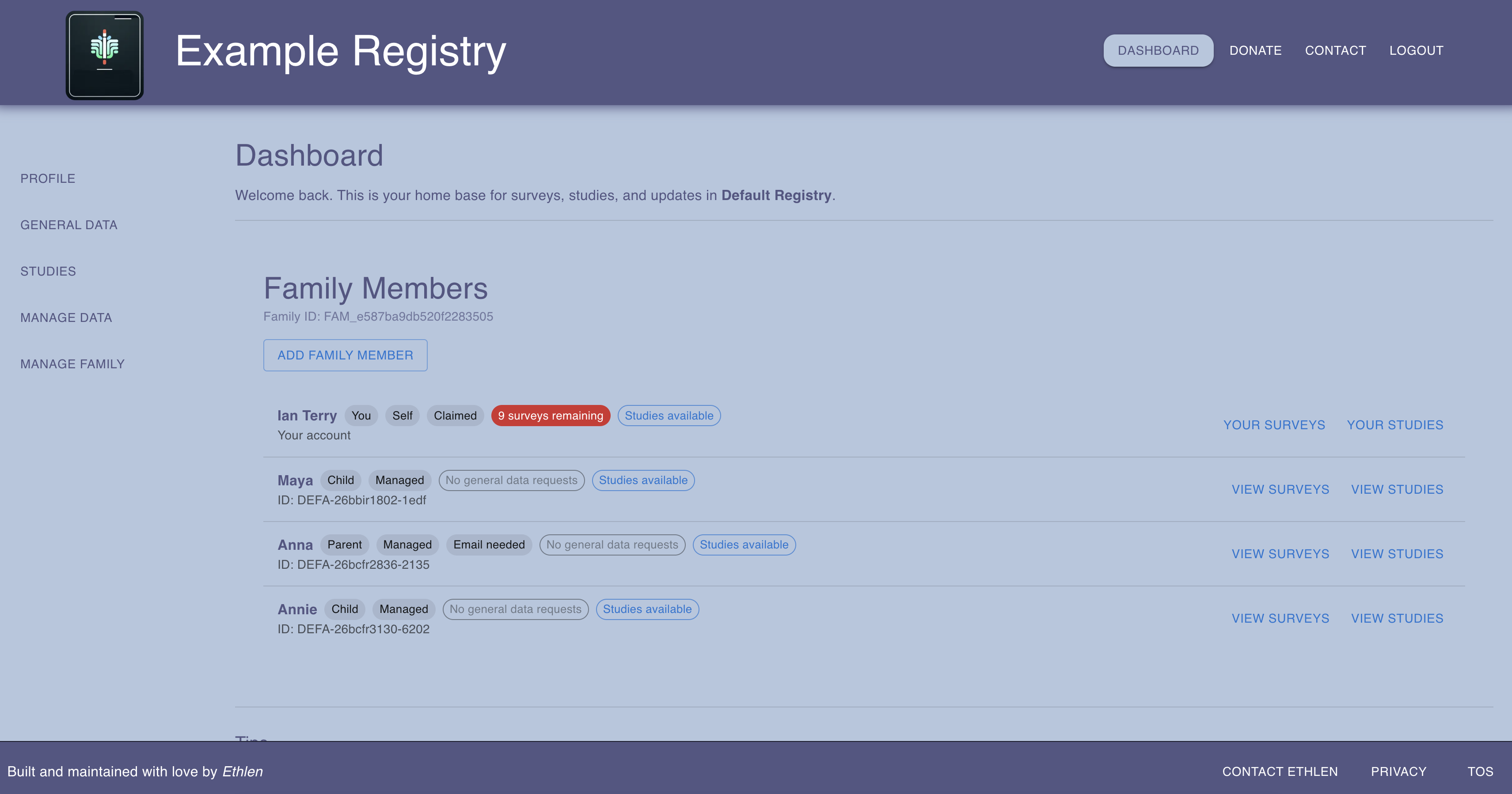
The participant dashboard — family management, survey tracking, and study enrollment in one place
What We Deliver
A fully branded, HIPAA and GDPR compliant participant portal — configured to your surveys, your consent language, and your workflows. No engineering team required on your end.
Your Data, Physically Isolated
Behind your portal sits the DigitalCabinet data infrastructure: a backend built around virtual private environments where your organization gets its own isolated storage, its own encryption keys, its own access controls, and its own origin validation. Your participants' data is physically and cryptographically separated from every other program on the platform.
What You Get
A Portal Built Around Your Program
Branded to your organization, accessible from any device. We configure your surveys, your consent language, and your study structure — you launch with a production-ready portal.
Flexible Enrollment Models
Self-serve registration, clinician-managed enrollment, or a hybrid. Some programs are fully participant-driven. Others run through care teams. We support all of it.
Study Enrollment & Management
Participants browse available studies, enroll, and withdraw. Each study carries its own scoped consent, tracked independently from portal participation.
Survey & Data Collection
Surveys configured directly into the platform. Responses flow into your private environment, tagged by participant, study, and respondent type.
File & Document Management
Participants and care teams upload, manage, and access health documents and records within your program's private vault.
Optional Payment Gating
For programs that charge participants for access, we integrate subscription billing into the enrollment flow. For programs that don't, it's simply not there.
How It Works
Three steps from kickoff to a live participant portal.
You Provide
Your branding, your surveys, your consent language, and your study structure.
We Configure & Deploy
Your portal — a fully isolated instance mapped to your organization's private environment.
You Launch
With a production-ready participant portal, optional clinician tooling, and a secure, compliant data backend.
Who This Is For
Academic medical centers running longitudinal patient cohorts
Biobanks building participant-facing data contribution programs
Disease foundations and advocacy organizations standing up research communities
Pharmaceutical and CRO teams managing participants across observational studies
Hospital networks running internal data collection programs
Ready to Launch Your Program?
Tell us about your research program, and we will show you how fast you can go live.

